Alzheimer’s disease, a neurodegenerative disorder, results in memory loss and compromises cognitive abilities in many people beyond the age of 60. Currently used techniques to detect manifestations of the disease (MRI, PET, and CT scans) are complex, expensive, and often produce inconclusive results.
“Our goal was to find a reliable, cost-effective solution,” says Debasis Das, Assistant Professor in the Department of Inorganic and Physical Chemistry (IPC), Indian Institute of Science (IISc). In a study published in Analytical Chemistry, he and Jagpreet Sidhu, a CV Raman postdoctoral fellow in IPC, have designed a small molecular fluorogenic probe that can sense a specific enzyme linked to the progression of Alzheimer’s disease. Such a probe can easily be fabricated into a strip-based kit that may enable on-site diagnosis.
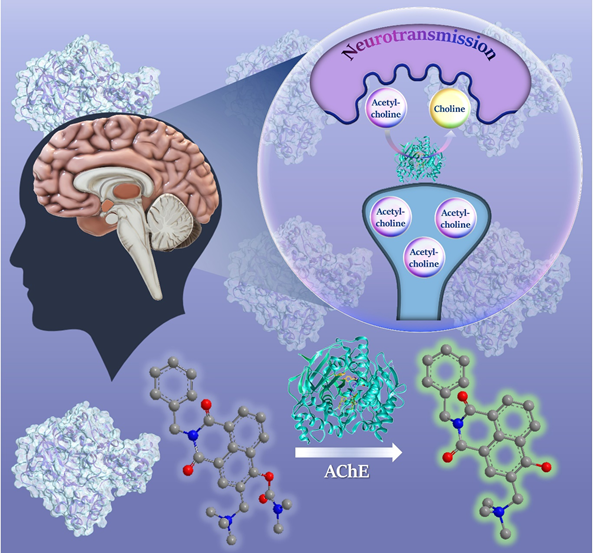
“Fluorogenic probes are not fluorescent by themselves, but upon reaction with a target enzyme, they become fluorescent,” explains Das. “Our target enzyme is Acetylcholinesterase (AChE).” Studies have shown that in the early stages of Alzheimer’s disease, AChE levels become imbalanced, thus making it a potential biomarker for the disease.
Brain cells or neurons secrete neurotransmitters – signalling molecules that instruct other cells to perform certain functions. Acetylcholine (ACh) is one such neurotransmitter; its levels in our nervous system are tightly controlled by enzymes like AChE, which breaks it down into two parts – acetic acid and choline. Current approaches determine AChE levels indirectly by measuring the levels of choline. They also often give confounding results because AChE has “sister enzymes” such as butyrylcholinesterase and cholinesterase that work on similar substrates, including ACh, says Das.
The team first analysed the crystal structures of the enzyme (AChE) and the substrate (ACh). Then, they designed a synthetic molecule that mimics ACh. The probe developed by the team has one structural element (quaternary ammonium) that interacts specifically with AChE, and another that binds to the active site in AChE and gets digested (just like natural ACh), giving out a fluorescent signal. The team tweaked the distance between the two elements to make it bind tightly to the enzyme. “In previous reports, people did not use this quaternary ammonium group. Because of this, they were not able to attain specificity and selectivity,” says Sidhu, who is the first author of the study.
To test the probe’s ability to be digested specifically by the enzyme, the team used commercially available AChE as well as lab-made human brain AChE expressed in bacteria. Although AChE has been extracted from the human brain, purified, and crystallised in the past, this is the first time that it has been purified in the active form after cloning and expressing it in a bacterial system, the researchers say.
In collaboration with Deepak Saini’s lab at the Department of Developmental Biology and Genetics, IISc, the team showed that the fluorogenic probe could also enter brain cells cultured in the lab and fluoresce upon contact with AChE.
“We now have a proof-of-concept and a lead. Our goal is to take it to translation, in an Alzheimer’s disease model. For this, we need to modify the probe,” says Das. Currently, the probe is UV-active, which can be harmful to tissues in high doses. “These modifications would lead to the development of near-infrared active probes, which would be safer for living cells and allow deep-tissue imaging. We are already quite close to doing this.”
Apart from Alzheimer’s disease, such a probe can also be used for other applications like detecting pesticide-related poisoning, as AChE can be inhibited by compounds used in some pesticides, Sidhu adds.