Cell division is fundamental for all life on Earth. One of the important steps in this process is anaphase. During anaphase, chromosomes that contain genes are equally distributed or segregated to two newly formed daughter cells. Chromosome segregation during anaphase is tightly coupled with cleavage furrow formation, bisecting the cells into two daughter cells. How chromosome segregation is coordinated with cleavage furrow formation in time and space is poorly understood.
Researchers at the Department of Microbiology and Cell Biology (MCB), IISc, led by Sachin Kotak, have found how certain essential protein complexes are distributed across two different regions in the cell membrane, and how they play a crucial role in coordinating chromosome segregation with cleavage furrow formation during anaphase.
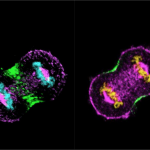
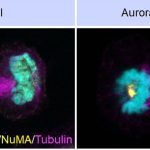
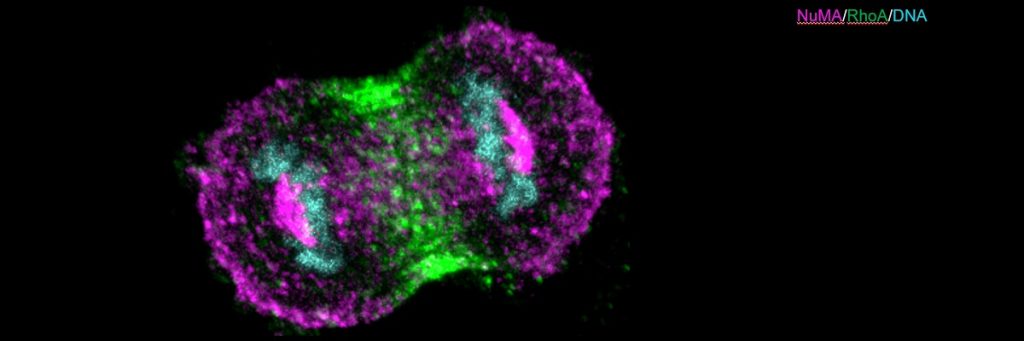
The team shows that two sets of protein complexes assemble at the cell membrane in a polarised manner, and each of these complexes antagonises the localisation of another complex. One of these is the Nuclear Mitotic Apparatus (NuMA) protein, together with the dynein/dynactin motor protein (referred to as NuMA-based complex). This complex is present in the polar region of the cell membrane, where it generates pulling forces on the spindle fibres to advance chromosome segregation. In contrast, the other complex made of Ect2/Cyk4/Mklp1 (referred to as Ect2-based complex) is restricted to the equatorial region of the cell membrane. This complex ensures that a small protein called RhoA accumulates at the equatorial membrane, which is essential for proper cleavage furrow formation.
The researchers carried out immunofluorescence and live-imaging analysis, and found that the Ect2-based complexes restrict NuMA-based complexes to the polar region, while the NuMA protein complexes, in turn, confine the Ect2-based proteins to the equatorial region.
This mutual restriction controls the localisation of dynein/dynactin at the polar region and RhoA at the equatorial region of the cell membrane, which ensures that chromosome segregation is coordinated with the cleavage furrow formation. The absence of such coordination will lead to genomic instability. The study, therefore, offers insights into the mechanisms that control key processes during cell division.