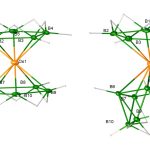
Water is generally regarded as the “matrix of life”. But how exactly does water help in biological reactions? This question has perplexed researchers for years. In recent work led by Biman Bagchi at the Solid State and Structural Chemistry Unit (SSCU), researchers have attempted to understand this issue by focusing on three different proteins: myoglobin, GB1 and dimeric insulin. In each of these systems, they considered a single reaction coordinate (hydrogen bond or coordination) and studied the effect of water on its dissociation. More specifically, they calculated the fiction components on the bond, separately from protein and water.
Their investigation reveals that the cross-correlation between the forces from these two domains results in a negative friction term at the reaction coordinate, which reduces the total friction and effectively increases the reaction rate. The action-at-a-distance of water on protein function and dynamics is shown to lead to a tug-of-war between internal (due to protein) and external (due to water) frictions, revealed clearly by considering the three different proteins. The researchers demonstrate that such out-of-phase behaviour in protein-water interactions, pointed out here for the first time, facilitates reactions through reduction of the friction on the reaction coordinate.